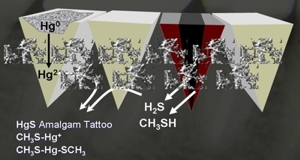
Bacterial toxins and amalgam
Root-treated teeth with neighbouring amalgam fillings are to be regarded as particularly critical. The mercaptan originating from the nonvital teeth derives its name from the juxtaposition of “mercurius” (the Latin name mercury) and “captans” for capturing. A marked propensity to absorb mercury is ascribed to this tooth toxin.
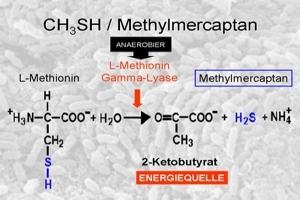
The metabolism of bacteria then converts this into methyl mercaptan, a flammable colourless fluid that is one of the strongest neurotoxins: just 0.1 millilitre, i.e. a ten thousandth of a litre, is lethal. The concentration in the mouth is much lower, but the long-term release of even the tiniest doses is not without its harmful effects on health. For example, dimethylmercury also penetrates the blood-brain barrier and accumulates in the brain.
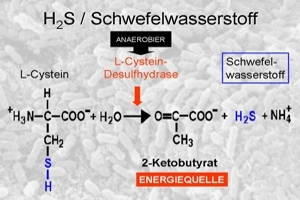
The synthesis is summarised in the following three diagrams:
Amalgam mercury reacts with the toxins from nonvital teeth to form even more toxic compounds.
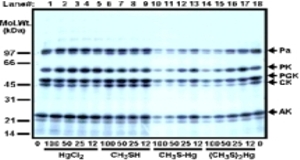
The toxic organic Hg compounds inhibit the radioactive marking of the test enzymes more strongly than HgCl2; this can be seen particularly clearly in the group on the right with methyl mercaptan.